Nitrogen Molar Mass: The Invisible Backbone of Chemistry’s Most Everyday Environment
Nitrogen Molar Mass: The Invisible Backbone of Chemistry’s Most Everyday Environment
Everywhere you look, chemistry is at work—fueling the air you breathe, forming the proteins in your body, and shaping the materials around you. Among the elements that quietly sustain these processes, nitrogen stands out: abundant yet often overlooked, its molar mass forming the silent backbone of countless everyday environments. With a molar mass of exactly 14.01 grams per mole, nitrogen is not the heaviest element on the periodic table, but its presence is anything but minor.
This article explores how nitrogen’s unique atomic identity underpins critical functions in air, agriculture, industry, and human biology—proving that chemistry’s most vital components often work unseen. Nitrogen’s Atomic Identity and Its Significance Nitrogen, symbolized as N, ranks as the seventh most abundant element in the universe and the fifth most prevalent in Earth’s crust and atmosphere. Its atomic number 7 reflects seven protons in the nucleus, with seven electrons in a stable outer shell.
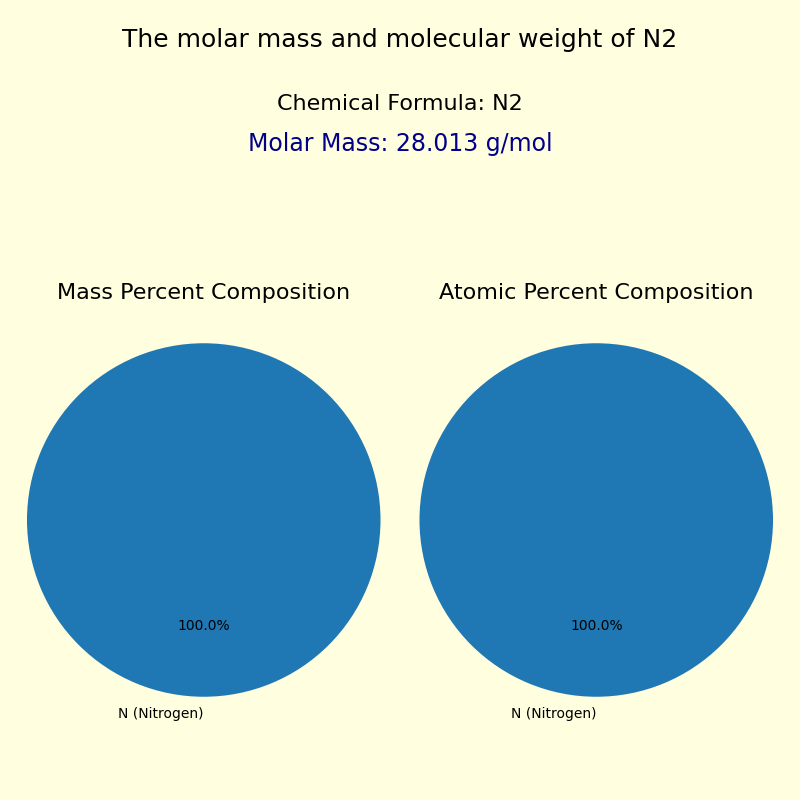
This configuration contributes to nitrogen’s relative chemical inertness under standard conditions—its full valence shell resists reaction, making it non-toxic and stable, which paradoxically allows it to persist and accumulate in vast quantities. The molar mass of nitrogen—14.01 g/mol—is derived from the sum of its isotopes, primarily nitrogen-14 (99.6%) and nitrogen-15 (0.4%). This precise value enables scientists to calculate exact quantities in chemical reactions, biological processes, and industrial applications.
“Understanding nitrogen’s molar mass is fundamental to modeling everything from atmospheric dynamics to fertilizer efficiency,” notes Dr. Elena Martinez, a chemical physicist specializing in environmental chemistry. “It’s the anchor point for measuring nitrogen’s role across scales.” Chemically, nitrogen’s light atomic weight grants it remarkable mobility: as dinitrogen gas (N₂), it freely diffuses through air, allowing rapid mixing yet maintaining a slow turnover rate—critical for natural cycles but challenging to alter efficiently.
This duality shapes nitrogen’s behavior in both biological and industrial spheres, where control over its reactivity is essential. Nitrogen as the Core of Earth’s Atmosphere Though nitrogen constitutes roughly 78% of Earth’s atmosphere by volume, its inertness often masks its profound influence. Unlike oxygen, which readily reacts to support respiration and combustion, nitrogen’s stable triply bonded diatomic molecule (N₂) resists biological degradation—requiring specialized enzymes, such as nitrogenase in bacteria, to convert it into usable forms like ammonia (NH₃) and nitrates (NO₃⁻).
This inertness, paradoxically, enables nitrogen’s indispensable role in sustaining life. The firm grasp of nitrogen’s molar mass allows researchers to track its global cycling—from atmospheric reservoirs to soil microorganisms, and from industrial fixation to oceanic absorption. “Without precise knowledge of nitrogen’s molar mass, we couldn’t model its fluxes or manage its environmental impacts effectively,” explains Dr.
Samuel Chen, a biogeochemist at the Global Nitrogen Research Initiative. “It’s the foundation of all nitrogen budgeting.” Nitrogen’s stability also supports its pervasive use in materials science. Synthetic polymers like nylon and polyester derive from nitrogen-rich monomers, their durability rooted in N–N and N–C bonds forged through careful manipulation of molecular architecture—architectures informed by molar mass precision.
Even in consumer electronics, nitrogen-based coolants and insulating gases leverage nitrogen’s non-reactive nature to protect sensitive components. Agricultural Dependence: The Nitrogen Cycle and Fertilizer Science Agriculture relies fundamentally on nitrogen, yet the element is biologically unavailable in its atmospheric form. Farmers depend on the nitrogen cycle—a complex, interconnected process where nitrogen transitions between air, soil, water, and living organisms—each stage calibrated by nitrogen’s molecular properties.
The synthesis of ammonia via the Haber-Bosch process revolutionized food production, converting atmospheric N₂ into reactive nitrogen compounds using high-pressure reactions driven by hydrogen and catalysts. “The Haber-Bosch process hinges on nitrogen’s molar mass,” says Dr. Liu Wei, an industrial chemist.
“By precisely controlling pressure and temperature, we overcome N₂’s inertness to yield ammonia—an essential building block for fertilizers.” However, nitrogen’s elementary stability also complicates sustainable use: excess fertilizers leach into waterways, fueling algal blooms and dead zones. “Understanding nitrogen’s molar mass helps design controlled-release fertilizers and catalytic converters that minimize waste,” notes Dr. Wei.
“It’s chemistry that enables smarter, cleaner agriculture.” In organic farming, nitrogen comes from natural sources like compost and leguminous plants, where microbial transformations convert atmospheric N₂ into plant-accessible forms. Yet even here, mastery of nitrogen’s molecular behavior—guided by its molar mass—remains critical to balancing yield with environmental stewardship. Industrial and Technological Applications From manufacturing to space propulsion, nitrogen’s unique properties drive innovation across sectors.
Its non-reactive nature makes it ideal as a protective atmosphere in food packaging, semiconductor fabrication, and electronic component storage—preventing oxidation and ensuring product integrity. In welding and cutting, nitrogen gas cools and shields molten metals, enhancing precision and strength. Industries such as metal fabrication and aerospace rely on nitrogen’s purity to maintain process consistency.
Energy systems also use nitrogen strategically. Liquid nitrogen chills superconducting materials and enables cryogenic testing. In clean energy research, nitrogen-based solvents and electrolytes are being explored for efficient hydrogen storage and fuel cell optimization.
“Every nitrogen application hinges on knowing its exact molar mass and molecular behavior,” states Dr. Miriam Torres, a chemical engineer at a leading materials lab. “It’s not just a number—it’s a control parameter that unlocks performance, safety, and sustainability.” The Invisible Backbone: Why Nitrogen Matters Everyday Beyond scientific laboratories, nitrogen’s influence permeates daily life.
Breathing clean air, eating nourishing food, wearing clothing made of synthetics—each trace nitrogen’s presence. Its molar mass, though small, enables scientific precision that underpins these invisible yet essential functions. “Nitrogen is chemistry’s quiet superstar,” observes Dr.
Elena Martinez again. “Its low but specific mass supports global ecosystems, alimentary systems, and industrial supply chains. Without this fundamental property, modern life as we know it would unravel.” In disinfecting medical tools, cooling reactives, and purifying gases, nitrogen’s purity and reliability, anchored in its atomic weight, become lifelines.
Its inertness guards organic compounds from premature oxidation, preserving medicines, fuels, and complex materials alike. Moreover, the growing urgency of climate change highlights nitrogen’s role in sustainable development. Strategies to reduce nitrogen pollution—from optimizing fertilizer use to capturing industrial emissions—depend on advanced understanding of nitrogen chemistry, driven by precise molar mass data.
Science in Motion: Tracking Nitrogen Through the Cycle
Scientists use nitrogen’s molar mass as a tracer across environmental and industrial systems. Isotopic analysis—measuring natural ratios of nitrogen-14 to nitrogen-15—reveals sources of pollution, migration pathways in ecosystems, and the origins of food products. + Understanding nitrogen cycling helps mitigate environmental damage.+ Tracking helps improve fertilizer efficiency in agriculture. + Industrial monitoring ensures safe handling and reduced waste. These applications grow more critical as urbanization and global food demand expand.
Precision in nitrogen measurement enables smarter regulation and innovation.
Balancing Abundance with Responsibility
While nitrogen’s overwhelming abundance might suggest ecological invulnerability, its atmospheric dominance masks a fragile cycle. Human activities have doubled global nitrogen fixation since the industrial era, straining natural balances.“Nitrogen’s molar mass reveals its resilience—but not its immunity,” warns Dr. Samuel Chen. “Using nitrogen swiftly must be matched by care: methods that reduce runoff, emissions, and energy use are essential.” Emerging technologies—bio-based fertilizers, carbon-nitrogen co-processes, and nitrogen-fixing crop engineering—promise a future where this invisible backbone supports growth without sacrificing planet health.
Final Reflections on Nitrogen’s Silent Power
Nitrogen, the life-giving gas composing most of our sky, remains one of chemistry’s quiet giants. Its molar mass of 14.01 grams per mole is more than a number—it is a keystone. It enables biological transformation, industrial precision, and environmental resilience.From the air we breathe to the materials we rely on, nitrogen’s presence is foundational, invisible, yet unmistakably vital. As science continues to unravel its full potential, nationalism of nitrogen’s role ensures that chemistry’s most humble element remains at the heart of everyday innovation.


Related Post

The Unstoppable Power of Strategic Storytelling: How Lori O'Quinn Reshapes Communications in the Modern Age

Hilton TMTP Employee: The Backbone of Hospitality Excellence
Kelsey Kushner Tied the Knot: A Marital Milestone Celebrated on August 21, 2021, in a Viral Youtube Homage

Behind the Brilliance: Unpacking the Talented Cast of Good Will Hunting

