What Visible Signs Reveal a Precipitation Reaction: The Science Behind the Spark in Tests
What Visible Signs Reveal a Precipitation Reaction: The Science Behind the Spark in Tests
When a chemical test yields a sudden burst of color, crystalline formation, or a startling visible spark, the cause often lies in a chain reaction known as precipitation—where soluble ions combine to form an insoluble solid. More than a mere spectacle, this transformation offers a window into the intricate chemistry governing solutions, guiding everything from lab diagnostics to environmental monitoring. What drives these striking changes, and why do they appear so visibly definitive?
The answer lies in the precise molecular interactions that trigger solid formation—and the telltale signs you can observe in the lab or field. This article explores the visible markers of precipitation reactions, decoding the science behind the spark that signals a chemical transformation.
The Molecular Dance: How Precipitation Reactions Unfold
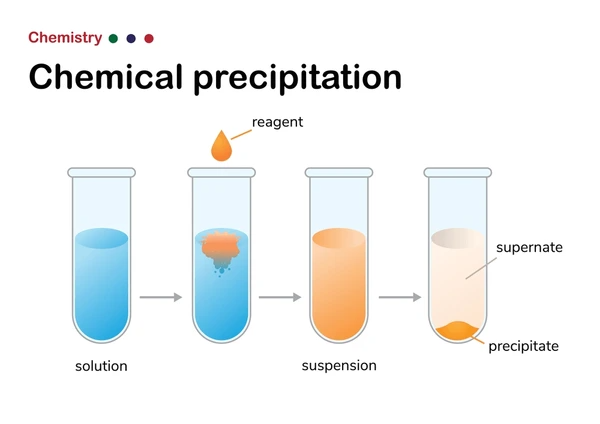
A precipitation reaction occurs when two aqueous solutions mix, and their ions combine to form an insoluble compound—precipitate—that exerts its own microbiome of visible effects.At the core, solubility rules govern these transformations: when the product of ion pairing exceeds a substance’s solubility limit, a solid precipitate emerges. This process is exothermic in many cases, sometimes releasing enough energy to cause observable thermal changes, though most visible signs stem from the rapid formation of a solid. Key indicators include: - Rapid color shifts—pale or clear solutions darkening sharply - Formation of colored or translucent particles ranging from powdery dusts to coarse crystals - Formation of a visible spark or fragment release, especially in solutions with reactive metal ions - Gas evolution that may contribute to bubbling or pressure, though less directly visible “Precipitation reactions are among the most immediate and recognizable forms of chemical change,” explains Dr.
Elena Marquez, analytical chemist at the National Institute of Standards. “The moment a precipitate becomes visible—whether through settling, color change, or physical agitation—it signals strong chemical binding.”
Color Shifts: The First Visual Clue to Chemistry in Action
One of the most immediate signs of a precipitation reaction is a distinct color change in the solution. As dissolved ions recombine into an insoluble compound, the environment shifts—altering electronic transitions that absorb light.This often manifests as deepening or shifting hues that betray the specific ions involved. For example, mixing barium nitrate with sodium carbonate produces a vivid white precipitate and instantly alters the solution’s tone; the precipitate remains immobile, while the altered chromatic profile permanently redefines the test medium. Other pairs yield equally dramatic visual effects: - Silver nitrate with chloride ions yields a dense, truccent black precipitate (AgCl), instantly recognizable due to both color and insolubility.
- In tests involving halide ions, chloride, bromide, and iodide each produce distinct precipitation patterns—chloride forms white flakes, bromide clouds pale yellow, and iodide yields pale violet crystals—serving as a visual shorthand recognized even in field conditions. These color shifts are more than aesthetic; they confirm ion pairing and insolubility, helping lab personnel pinpoint components rapidly.
Texture and Morphology: From Cloudiness to Crystalline Order
Visible changes extend beyond color into texture: a clear solution may cloud abruptly as particle nuclei form, then settle.This cloudiness—turbidity—results from light scattering by suspended particles too small to sediment immediately but large enough to disrupt transmission. More striking is the evolution into defined crystals. When conditions favor growth over mere precipitation, delicate geometries emerge— Needle-like silver chloride, cubic lead sulfide, or hexagonal barium sulfate crystals each form distinctive textures that signal specific reaction kinetics.
Observing such morphology helps confirm not just presence but maturity of precipitation. “Instability in the reaction produces fine, amorphous gels first, evolving into ordered crystals as equilibrium is reached,” notes Dr. Marquez.
“This progression reveals underlying thermodynamics.” High-resolution imaging in educational labs and industrial quality control now routinely captures precipitate morphology, turning the visual into quantitative data.
The Spark of Reaction: When Precipitation Meets Energy and Momentum
Perhaps the most dramatic visible sign is a sudden spark or violent release—especially in reactions involving highly reactive metal ions. When solutions containing radium or uranium salts meet reagents like ammonium carbonate or hydrogen peroxide in controlled conditions, rapid precipitation can generate micro-scale sparks or crystalline ejections.These sparks, often overlooked, represent kinetic energy release from the sudden formation of stable solids under electrostatic and lattice-energy constraints. Although not universal—many precipitation reactions proceed silently—the spark remains a powerful indicator in systems where redox imbalances or ionic charge reconciliation trigger instant solidification. The bright, fleeting spark is a micro-event mirroring the macroscopic bond formation, visually anchoring the reaction’s exothermic signature.
What matters most is not mere spectacle, but what these signals reveal: confidence in identification, transparency in monitoring, and precision in understanding chemical environments. From the sudden darkening of a carbonate solution to the glitter of silver chloride crystallizing in a tube, visible signs of precipitation reactions serve as both scientific evidence and storytelling devices within chemistry. They bridge abstract molecular interactions with tangible observation, allowing chemists, students, and engineers to detect, verify, and interpret reactions with clarity. With proper training, these visual cues transform complex ion behavior into actionable knowledge—ensuring accuracy in research, diagnostics, and applied science.
As analytical methods advance, the science behind the spark grows ever more precise, reaffirming that even the most visible chemical transformations are rooted in deep, predictable physics and chemistry.


/precipitation-reaction-when-adding-lead-nitrate-to-pottasium-iodine-to-form-lead-iodine-as-yellow-precipitate-in-bottle-131985882-58ea34a53df78c5162f899a7.jpg)
Related Post

Ll Cool J’s grandson turns 5, marking a milestone in hip-hop legacy and family tradition

Did Awkwafina Ever Be Involved in a Relationship? Unveiling Her Romantic History
Pokeland Legends Codes 2024: Working Codes & Reddit Updates

Roaming Hotspot: Can Your Overseas Data Share? What You Need to Know Before Connecting Abroad

